MGC Pharma expands presence in budding UK cannabis market
Published 08-AUG-2018 12:41 P.M.
|
2 minute read
Hey! Looks like you have stumbled on the section of our website where we have archived articles from our old business model.
In 2019 the original founding team returned to run Next Investors, we changed our business model to only write about stocks we carefully research and are invested in for the long term.
The below articles were written under our previous business model. We have kept these articles online here for your reference.
Our new mission is to build a high performing ASX micro cap investment portfolio and share our research, analysis and investment strategy with our readers.
Click Here to View Latest Articles
Specialist pot-stock, MGC Pharmaceuticals (ASX:MXC), revealed this morning that luxury British department store, Harvey Nichols, has expanded the sale of the premium cannabidiol (CBD) MGC Derma and Derma Plus collections to two further stores within the UK — Birmingham and Edinburgh.
This expansion comes from strong and increasing demand following the overwhelming success of the launch at the Knightsbridge store in the June 2018 quarter. Revenue is expected to increase with these additional two stores, with sales driven further by a razor-sharp PR and marketing campaign that has positively impacted the MGC Derma footprint within the UK.

Of course, as always, success is not guaranteed — consider your own personal circumstances before investing, and seek professional financial advice.
The ongoing campaign, which has targeted press and social influencers, has enabled considerable brand exposure for MGC Derma across a range of prestigious (and far-reaching) print and online publications.
These include The Guardian, which has a circulation of 7.7 million, The Daily Mail (circulation 20.1 million), The Evening Standard (circulation 11.7 million) and BritishBeautyBlogger.com — circulation 30,000, and is directly linked to increasing brand awareness and sales. A taste of MXC in the media:
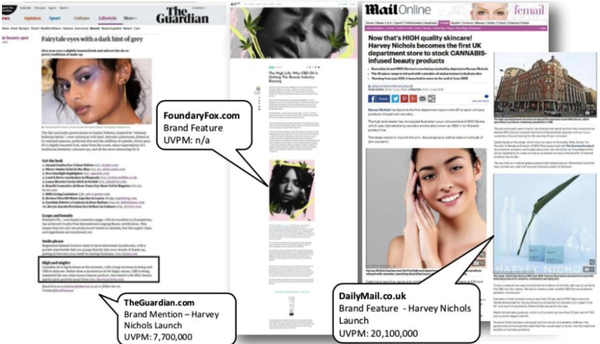
With its ever-expanding footprint and sizeable network of relationships, there’s a clear gateway for MGC Pharma into the UK market. Following recent changes in UK legislation on the use of medicinal cannabis, MGC Derma’s growing presence in the UK should provide MGC Pharma with a competitive edge in accessing this nascent market.
In a separate milestone altogether, three of MXC’s research-backed premium CBD products within the MGC Derma AntiAging collection have also been shortlisted for the Notebook Anti-Aging Beauty Awards. This makes for a significant achievement, validating the efficacy of MXC’s innovative products.
Roby Zomer, MGC Pharma co-founder and Managing Director, commented: “I am delighted to report that the Harvey Nichols’ launch of our MGC Derma and Derma Plus collections has been a success and is extending the products lines to two more stores. We have focused a lot of attention on building our brand exposure and presence in the UK market and are pleased to see it has paid off.”
“The UK is an exciting market for us due to recent changes in legislation and I look forward to exploring the possibilities for the MGC pharma division here.”
This latest development comes hot on the heels of a wave of positive newsflow from MXC. Last month, the bio-pharma small cap smashed a milestone of the highest order, securing full Good Manufacturing Practice (GMP) certification for its European pot production and compounding facility — the final step towards kicking off a commercial-scale production operation.
General Information Only
S3 Consortium Pty Ltd (S3, ‘we’, ‘us’, ‘our’) (CAR No. 433913) is a corporate authorised representative of LeMessurier Securities Pty Ltd (AFSL No. 296877). The information contained in this article is general information and is for informational purposes only. Any advice is general advice only. Any advice contained in this article does not constitute personal advice and S3 has not taken into consideration your personal objectives, financial situation or needs. Please seek your own independent professional advice before making any financial investment decision. Those persons acting upon information contained in this article do so entirely at their own risk.
Conflicts of Interest Notice
S3 and its associated entities may hold investments in companies featured in its articles, including through being paid in the securities of the companies we provide commentary on. We disclose the securities held in relation to a particular company that we provide commentary on. Refer to our Disclosure Policy for information on our self-imposed trading blackouts, hold conditions and de-risking (sell conditions) which seek to mitigate against any potential conflicts of interest.
Publication Notice and Disclaimer
The information contained in this article is current as at the publication date. At the time of publishing, the information contained in this article is based on sources which are available in the public domain that we consider to be reliable, and our own analysis of those sources. The views of the author may not reflect the views of the AFSL holder. Any decision by you to purchase securities in the companies featured in this article should be done so after you have sought your own independent professional advice regarding this information and made your own inquiries as to the validity of any information in this article.
Any forward-looking statements contained in this article are not guarantees or predictions of future performance, and involve known and unknown risks, uncertainties and other factors, many of which are beyond our control, and which may cause actual results or performance of companies featured to differ materially from those expressed in the statements contained in this article. S3 cannot and does not give any assurance that the results or performance expressed or implied by any forward-looking statements contained in this article will actually occur and readers are cautioned not to put undue reliance on forward-looking statements.
This article may include references to our past investing performance. Past performance is not a reliable indicator of our future investing performance.






