Creso delivers robust year-on-year revenue growth
Hey! Looks like you have stumbled on the section of our website where we have archived articles from our old business model.
In 2019 the original founding team returned to run Next Investors, we changed our business model to only write about stocks we carefully research and are invested in for the long term.
The below articles were written under our previous business model. We have kept these articles online here for your reference.
Our new mission is to build a high performing ASX micro cap investment portfolio and share our research, analysis and investment strategy with our readers.
Click Here to View Latest Articles
Shares in Creso Pharma Limited (ASX:CPH, FRA:1X8) soared 20% on Friday after the company released its quarterly report for the three months to 31 December, 2019.
Momentum has been building since early December as the company outlined its strategy for generating substantial growth in 2020.
This has triggered a share price increase of 50% since early December.
The quarterly report featured strong revenue growth, as well as highlighting promising operational developments within the group.
Unaudited consolidated group revenues grew very strongly in the December quarter, reaching nearly $2 million, and bringing full year revenues to $3.6 million for 2019.
Year-on-year revenue boosted by Creso Pharma’s European nutraceutical and animal health product sales during the December quarter was nearly $1.6 million.
Equivalent unaudited revenues from nutraceutical and animal health product sales for the full year 2019 were nearly $2.8 million, up from about $550,000 for the full year 2018.
cannaQIX® 50 launched in Australia
Creso Pharma also made substantial ground on the operational front with its broad range of products.
The company delivered its first orders of medicinal 10% CBD oil in New Zealand.
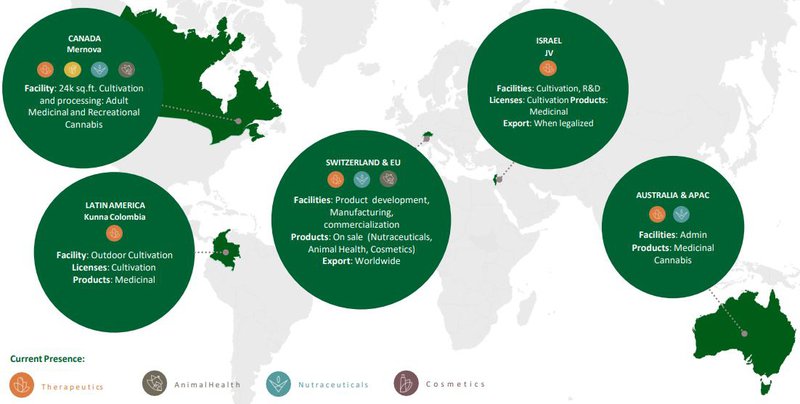
There was also good news in relation to the group’s flagship medical cannabis product cannaQIX® 50 with sales commencing in Australia and the launch of the cannaQIX® range in South Africa anticipated in April 2020.

The company has partnered with leading South African pharmaceutical company Pharma Dynamics in South Africa, and it has placed two initial orders for cannaQIX® Regular, totalling approximately AU$300,000.
The partnership will also pursue commercialisation of Creso Pharma’s products in other African countries, including Angola, Botswana, Lesotho, Namibia, Mozambique, Swaziland, Uganda and Zimbabwe.
Mernova ramping up to full production
The 24,000 square foot world-class, revenue generating Mernova Cultivation Facility is in the advanced stages of securing European Union GMP certification, which will allow the export of its GMP medicinal cannabis products to Europe.
Upon receiving this certification, Mernova will be one of few Canadian companies with a licence to export to the EU.
Mernova’s unaudited revenues for the full year 2019 were $877,000 in its first year of production.
The facility commenced production midway through 2019 and has been scaling up. The facility is not yet at full production capacity, which is anticipated to occur during 2020.
The granting of a licence in February to cultivate at Creso Pharma’s “Mernova Medical” facility in Nova Scotia made Creso the only ASX listed company that has 100% ownership of a Canadian licensed producer.
Product pipeline
Creso Pharma currently has a portfolio of 13 products which have been developed with standardised dosing and formulations through the application of pharmaceutical rigour, GMP standards and innovative delivery technologies.
Of the 13, four have been commercialised, with nine pending commercialisation.
During the December quarter, Creso Pharma continued the protection of its sustainable competitive advantage with the conclusion of the international phase of the PCT and the commencement of national filing in all European countries, Australia, New Zealand, South Africa and selected North and South American countries for both the human health and the animal health delivery systems.
The company is well-positioned to fund its growth initiatives in 2020 with $2.8 million in cash as at 31 December, 2019.
Management also noted that the company has undrawn debt facilities of $4.2 million.
Commenting on Creso Pharma’s outlook, chief executive and co-founder Dr Miri Halperin Wernli, said, ‘’Creso Pharma has an expanding portfolio of innovative products and a leadership team with vast experience in the pharmaceutical industry.
"The company’s products are now sold across Europe and Oceania, and will soon be available in South Africa.
‘’We are confident in our ability to successfully commercialise our new and existing products, and we look forward to updating shareholders on our continuing progress and growth.”
General Information Only
S3 Consortium Pty Ltd (S3, ‘we’, ‘us’, ‘our’) (CAR No. 433913) is a corporate authorised representative of LeMessurier Securities Pty Ltd (AFSL No. 296877). The information contained in this article is general information and is for informational purposes only. Any advice is general advice only. Any advice contained in this article does not constitute personal advice and S3 has not taken into consideration your personal objectives, financial situation or needs. Please seek your own independent professional advice before making any financial investment decision. Those persons acting upon information contained in this article do so entirely at their own risk.
Conflicts of Interest Notice
S3 and its associated entities may hold investments in companies featured in its articles, including through being paid in the securities of the companies we provide commentary on. We disclose the securities held in relation to a particular company that we provide commentary on. Refer to our Disclosure Policy for information on our self-imposed trading blackouts, hold conditions and de-risking (sell conditions) which seek to mitigate against any potential conflicts of interest.
Publication Notice and Disclaimer
The information contained in this article is current as at the publication date. At the time of publishing, the information contained in this article is based on sources which are available in the public domain that we consider to be reliable, and our own analysis of those sources. The views of the author may not reflect the views of the AFSL holder. Any decision by you to purchase securities in the companies featured in this article should be done so after you have sought your own independent professional advice regarding this information and made your own inquiries as to the validity of any information in this article.
Any forward-looking statements contained in this article are not guarantees or predictions of future performance, and involve known and unknown risks, uncertainties and other factors, many of which are beyond our control, and which may cause actual results or performance of companies featured to differ materially from those expressed in the statements contained in this article. S3 cannot and does not give any assurance that the results or performance expressed or implied by any forward-looking statements contained in this article will actually occur and readers are cautioned not to put undue reliance on forward-looking statements.
This article may include references to our past investing performance. Past performance is not a reliable indicator of our future investing performance.






