Cashed up Imugene to present at American Association for Cancer Research
Hey! Looks like you have stumbled on the section of our website where we have archived articles from our old business model.
In 2019 the original founding team returned to run Next Investors, we changed our business model to only write about stocks we carefully research and are invested in for the long term.
The below articles were written under our previous business model. We have kept these articles online here for your reference.
Our new mission is to build a high performing ASX micro cap investment portfolio and share our research, analysis and investment strategy with our readers.
Click Here to View Latest Articles
Clinical stage immuno-oncology company, Imugene Limited (ASX:IMU), is developing a range of new and novel immunotherapies that seek to activate the immune system of cancer patients to treat and eradicate tumors.
Its unique platform technology seeks to harness the body’s immune system to generate antibodies against tumours, potentially achieving a similar or greater effect than synthetically manufactured monoclonal antibody therapies.
Product pipeline includes multiple immunotherapy B-cell vaccine candidates aimed at treating a variety of cancers in combination with standard of care drugs and emerging immunotherapies.
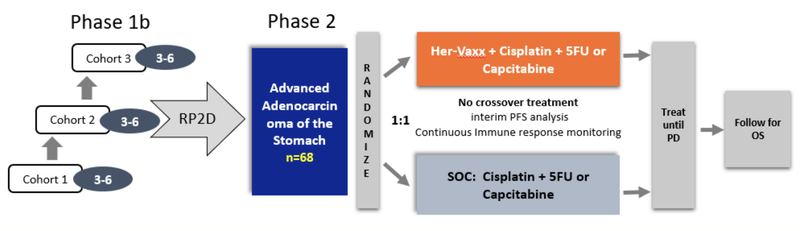
Imugene’s Phase 1b/2 clinical trial is targeting patients with HER-2 positive gastric cancer. The group selected HER-2 positive gastric cancer as this type is not nearly as well served as breast cancer, yet still has approximately the same number of patients being HER-2 positive and is more severe than breast cancer, offering a significant market opportunity for HER-Vaxx.
The Phase 1b stage of the Phase 1b/2 trial has completed with all endpoints being met. Phase 1b established safety, tolerability and the dose for the Phase 2 portion of the trial.
The Phase 2 stage of the HER-Vaxx trial in HER-2 positive gastric cancer commenced in February 2019.
The company today provided a half year review of its financial results to December 31, 2018.
The group reported a loss for the half-year ended 31 December 2018 of $3,449,097. This was up from $1,632,852 a year earlier largely due to the significant increase in clinical trial and research activities undertaken by the group.
On the back of a successful $20.1 million capital raise (before costs) in July 2018, the group's net assets increased to $31,189,570 compared with $15,475,479 at 30 June 2018. As at 31 December 2018, the group had cash reserves of $24,053,140, up from $7,822,057 at (30 June 2018).
The focus of the group’s operations in the short- to medium-term will be directed at the Phase 1b/2 gastric cancer study design, as follows:
- HER-Vaxx: recruitment of phase 2 trial with gastric cancer patients,
- KEY-Vaxx: accelerate pre-clinical studies; obtain an IND ‘go’ and to commence the clinical trial.
The company also announced that Professor Ursula Wiedermann will present on the HER-Vaxx cancer vaccine at the American Association for Cancer Research (AACR) 2019 Annual Meeting scheduled for March 29 to April 3, 2019.
The poster will be presented by Prof. Wiedermann, the co-inventor of the HER-Vaxx cancer vaccine and member of the IMU’s Scientific Advisory Board.
The abstract presentation is titled ‘A Phase Ib open label multicenter study with a HER2/neu peptide vaccine administered with cisplatin and 5-fluorouracil or capecitabine chemotherapy shows safety, immunogenicity and clinical response in patients with HER2/Neu overexpressing advanced cancer of the stomach.’
Imugene’s HER-Vaxx is a B-cell peptide cancer vaccine designed to treat tumours that over-express the HER-2/neu receptor, such as gastric, breast, ovarian, lung and pancreatic cancers. The immunotherapy is constructed from several B cell epitopes derived from the extracellular domain of HER-2/neu. It has been shown in pre-clinical studies and in Phase I studies to stimulate a potent polyclonal antibody response to HER-2/neu, a well-known and validated cancer target.
General Information Only
S3 Consortium Pty Ltd (S3, ‘we’, ‘us’, ‘our’) (CAR No. 433913) is a corporate authorised representative of LeMessurier Securities Pty Ltd (AFSL No. 296877). The information contained in this article is general information and is for informational purposes only. Any advice is general advice only. Any advice contained in this article does not constitute personal advice and S3 has not taken into consideration your personal objectives, financial situation or needs. Please seek your own independent professional advice before making any financial investment decision. Those persons acting upon information contained in this article do so entirely at their own risk.
Conflicts of Interest Notice
S3 and its associated entities may hold investments in companies featured in its articles, including through being paid in the securities of the companies we provide commentary on. We disclose the securities held in relation to a particular company that we provide commentary on. Refer to our Disclosure Policy for information on our self-imposed trading blackouts, hold conditions and de-risking (sell conditions) which seek to mitigate against any potential conflicts of interest.
Publication Notice and Disclaimer
The information contained in this article is current as at the publication date. At the time of publishing, the information contained in this article is based on sources which are available in the public domain that we consider to be reliable, and our own analysis of those sources. The views of the author may not reflect the views of the AFSL holder. Any decision by you to purchase securities in the companies featured in this article should be done so after you have sought your own independent professional advice regarding this information and made your own inquiries as to the validity of any information in this article.
Any forward-looking statements contained in this article are not guarantees or predictions of future performance, and involve known and unknown risks, uncertainties and other factors, many of which are beyond our control, and which may cause actual results or performance of companies featured to differ materially from those expressed in the statements contained in this article. S3 cannot and does not give any assurance that the results or performance expressed or implied by any forward-looking statements contained in this article will actually occur and readers are cautioned not to put undue reliance on forward-looking statements.
This article may include references to our past investing performance. Past performance is not a reliable indicator of our future investing performance.