BARD1 releases further positive results
Published 06-MAR-2018 12:36 P.M.
|
2 minute read
Hey! Looks like you have stumbled on the section of our website where we have archived articles from our old business model.
In 2019 the original founding team returned to run Next Investors, we changed our business model to only write about stocks we carefully research and are invested in for the long term.
The below articles were written under our previous business model. We have kept these articles online here for your reference.
Our new mission is to build a high performing ASX micro cap investment portfolio and share our research, analysis and investment strategy with our readers.
Click Here to View Latest Articles
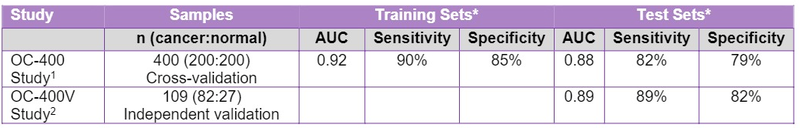
BARD1 Life Sciences (ASX:BD1) has announced additional positive results from its BARD1-Ovarian test, indicating that the test returned 89% sensitivity and 82% specificity.
BD1 conducted the follow on OC-400V study to assess the effectiveness of its ovarian algorithm generated in the OC-400 study to detect ovarian cancer in an independent test of 82 new ovarian cancers and 27 previously tested healthy controls.
The results of the OC-400V study (as indicated below) are highly promising and showcase high accuracy for detection of ovarian cancer. The results are an improvement over those recorded in the OC-400 study of 82% sensitivity and 79% specificity in cross validation tests.
Thanks to the results, BD1 confirmed the promise of its algorithm and now believes that BARD1-Ovarian has the potential to become a key diagnostic aid for early detection of the cancer.
It should be noted, however, that this is an early stage company and success is no guarantee. Investors should seek professional financial advice before making an investment.
BD1 Executive Director and CSO Dr Irmgard Irminger-Finger spoke on the promising results, “This study provides further evidence of the robustness of the BARD1-Ovarian test.
“Upon transfer to a commercial platform, our ongoing product development efforts will focus on testing BARD1-Ovarian in larger sample sets and the addition of other immunogenic markers to our biomarker panel to further train the algorithm and increase its accuracy for early detection of ovarian cancer.”
BD1 CEO Leearne Hinch also informed the market that the company was in discussions with a number of contract laboratory organisations that could transfer its research essay to a commercial platform.
“BARD1 anticipates that transfer of our research assay to a commercial platform will enable more efficient development and commercialisation of our diagnostic pipeline for detection of lung, ovarian and other cancers,” she said.
General Information Only
S3 Consortium Pty Ltd (S3, ‘we’, ‘us’, ‘our’) (CAR No. 433913) is a corporate authorised representative of LeMessurier Securities Pty Ltd (AFSL No. 296877). The information contained in this article is general information and is for informational purposes only. Any advice is general advice only. Any advice contained in this article does not constitute personal advice and S3 has not taken into consideration your personal objectives, financial situation or needs. Please seek your own independent professional advice before making any financial investment decision. Those persons acting upon information contained in this article do so entirely at their own risk.
Conflicts of Interest Notice
S3 and its associated entities may hold investments in companies featured in its articles, including through being paid in the securities of the companies we provide commentary on. We disclose the securities held in relation to a particular company that we provide commentary on. Refer to our Disclosure Policy for information on our self-imposed trading blackouts, hold conditions and de-risking (sell conditions) which seek to mitigate against any potential conflicts of interest.
Publication Notice and Disclaimer
The information contained in this article is current as at the publication date. At the time of publishing, the information contained in this article is based on sources which are available in the public domain that we consider to be reliable, and our own analysis of those sources. The views of the author may not reflect the views of the AFSL holder. Any decision by you to purchase securities in the companies featured in this article should be done so after you have sought your own independent professional advice regarding this information and made your own inquiries as to the validity of any information in this article.
Any forward-looking statements contained in this article are not guarantees or predictions of future performance, and involve known and unknown risks, uncertainties and other factors, many of which are beyond our control, and which may cause actual results or performance of companies featured to differ materially from those expressed in the statements contained in this article. S3 cannot and does not give any assurance that the results or performance expressed or implied by any forward-looking statements contained in this article will actually occur and readers are cautioned not to put undue reliance on forward-looking statements.
This article may include references to our past investing performance. Past performance is not a reliable indicator of our future investing performance.



