The US$11B global vape market is ablaze with innovation
Published 12-NOV-2018 11:36 A.M.
|
4 minute read
Hey! Looks like you have stumbled on the section of our website where we have archived articles from our old business model.
In 2019 the original founding team returned to run Next Investors, we changed our business model to only write about stocks we carefully research and are invested in for the long term.
The below articles were written under our previous business model. We have kept these articles online here for your reference.
Our new mission is to build a high performing ASX micro cap investment portfolio and share our research, analysis and investment strategy with our readers.
Click Here to View Latest Articles
As the legal weed market matures, it’s quickly driving advances in cannabis technologies, especially in terms of developing easier, safer and smarter ways to ingest.
One of the fastest growing segments in the cannabis industry is the seemingly humble vaporiser, which in turn is helping to create new markets for legalised weed.
According to a report by BIS Research, the global recreational and medicinal vapor market was estimated at US$11.43 billion in 2016 and is expected to grow over US$86.43 billion by 2025.
“Vaping” commonly refers to a method of cannabis consumption where the user inhales “vapor” or aerosol. To create this vapor, the active compounds in cannabis are heated at low temperatures. Unlike what happens when pot is smoked, no combustion or burning takes place at these lower temperatures.
This quick-acting and relatively discreet way to medicate (or to just have a good time, if that’s your jam) is generally considered safer and healthier than smoking.
Today’s vape market includes e-cigarettes, vape pens and advanced personal vaporisers (otherwise known as “mods”). Vape cartridges are filled with cannabis oil extracted from the plant.
A report by BDS Analytics and Arcview Market Research projects that nearly 60% of extract (or concentrate) spending in 2018 will be for prefilled vaporisers.
Spending on vape is expected to equal or perhaps surpass spending on flower by 2022.
This kind of intense growth, in turn, means that a lot of money is being funnelled into ways to improve the vape delivery system.
One company that’s targeting this growing market is medical diagnostic and monitoring technology developer, Lifespot Health (ASX:LSH).
Approaching the vaping space from a medical perspective (at least, for now), Lifespot has developed a vaporiser embedded with certified software infrastructure.
The company has a first-to-market advantage with the integrated Seng-Vital Cannamed® Bluetooth cannabis vaporising system, which provides a straight-forward, secure and self-controlled method to deliver medicines.
Lifespot subsidiary, Seng Vital, developed the vaporiser, which is activated by a patient’s fingerprint. It can then be used to inhale cannabis from oils or herbs.
The monitoring and collection software, in turn, gathers and collates data and readings from the vaporiser, which patients and their authorised physicians can then review and evaluate.

Something else that makes the vaporiser unique is its embedment in the medically certified software infrastructure of BodyTel GmbH — another Lifespot subsidiary.
This infrastructure allows both patients and authorised physicians to retrieve and evaluate the readings and data collected by the vaporiser.
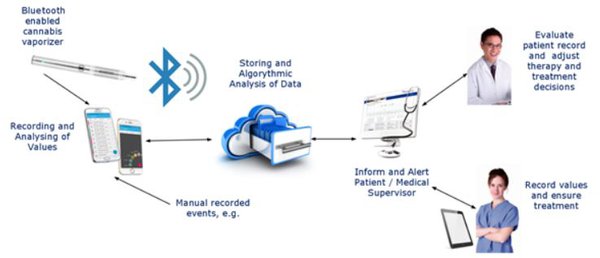
This integrated system isn’t restricted to vaping – it can also input data from edibles and drinks, which opens the company up to a number of significant global pot opportunities.
The method of inhalation used with the smart vaporiser, in turn, provides a number of clinical advantages. This system is especially well-suited to patient populations who are unable to swallow tablets, or for medications that are extensively metabolised prior to entry into the bloodstream.
“Studies tell us that oral forms of cannabis-based therapeutics have been shown to have low bioavailability following oral administration, so the dose required may be considerably higher than required for inhalation,” said Lifespot chairman, Rob Hannington. “Accordingly, the potential use of a lower dose as an inhaled format may in turn have a safety benefit.”
“Our integrated hardware and quality management platform provides a safe, measurable and controllable method for vaporising cannabis oil.”
Lifespot has demonstrated the benefits of this vaping system to a number of major pot players across Canada, USA, Israel, Australia and New Zealand — markets that are considerably advanced in approvals for both medical and recreational weed. Lifespot is also well-positioned for the next wave of approvals pending in European countries.
“We understand from our meetings that we are first to market a delivery device with these unique feature, and the major cannabis players see great value in our unique assets that are real enablers for them to register and commercialise their cannabis vaporising products,” Hannington said.
General Information Only
S3 Consortium Pty Ltd (S3, ‘we’, ‘us’, ‘our’) (CAR No. 433913) is a corporate authorised representative of LeMessurier Securities Pty Ltd (AFSL No. 296877). The information contained in this article is general information and is for informational purposes only. Any advice is general advice only. Any advice contained in this article does not constitute personal advice and S3 has not taken into consideration your personal objectives, financial situation or needs. Please seek your own independent professional advice before making any financial investment decision. Those persons acting upon information contained in this article do so entirely at their own risk.
Conflicts of Interest Notice
S3 and its associated entities may hold investments in companies featured in its articles, including through being paid in the securities of the companies we provide commentary on. We disclose the securities held in relation to a particular company that we provide commentary on. Refer to our Disclosure Policy for information on our self-imposed trading blackouts, hold conditions and de-risking (sell conditions) which seek to mitigate against any potential conflicts of interest.
Publication Notice and Disclaimer
The information contained in this article is current as at the publication date. At the time of publishing, the information contained in this article is based on sources which are available in the public domain that we consider to be reliable, and our own analysis of those sources. The views of the author may not reflect the views of the AFSL holder. Any decision by you to purchase securities in the companies featured in this article should be done so after you have sought your own independent professional advice regarding this information and made your own inquiries as to the validity of any information in this article.
Any forward-looking statements contained in this article are not guarantees or predictions of future performance, and involve known and unknown risks, uncertainties and other factors, many of which are beyond our control, and which may cause actual results or performance of companies featured to differ materially from those expressed in the statements contained in this article. S3 cannot and does not give any assurance that the results or performance expressed or implied by any forward-looking statements contained in this article will actually occur and readers are cautioned not to put undue reliance on forward-looking statements.
This article may include references to our past investing performance. Past performance is not a reliable indicator of our future investing performance.

